Medical
We have been engaged in the development, manufacturing, and certification of medical devices, including all related processes in accordance with ISO 13485, since 2004. In 2025, we successfully certified and implemented ISO 27001 for information and cybersecurity.
We are among the European leaders in telemedicine, e-health, and services for healthcare facilities. We provide customers with a comprehensive service—from the development and production of selected medical devices to custom software development and certification according to the MDR.
We are a member of the Association of Manufacturers and Suppliers of Medical Devices.
-
21 years
industry experience
-
6
products for healthcare
-
10+
countries with ZAT supplies
-
2023
Medical Device Regulation (MDR) certification
Healthcare products
Medical devices
We are a certified manufacturer of medical devices with extensive know-how in the development and production of medical devices intended for therapeutic purposes. Our medical devices are successfully used in patient treatment worldwide.
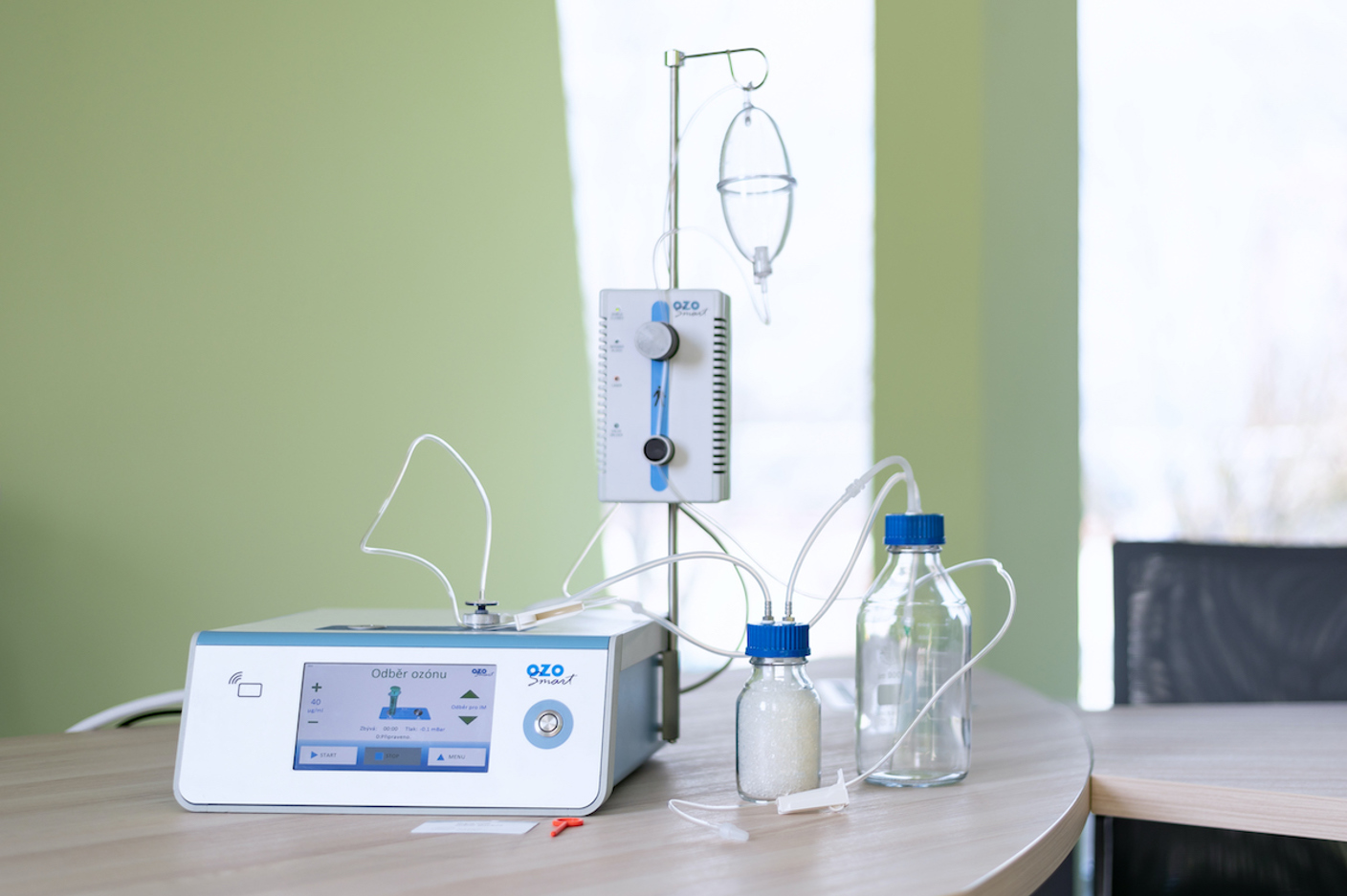
As the sole manufacturer of ozone therapy devices in the Czech Republic, we are committed to raising awareness about the benefits and importance of ozone therapy for patients.
Ozonotherapy
Ozosmart – an ozone therapy device – is an active therapeutic medical device designed for ozone application in clinical and outpatient practice and is an appropriate response to the needs of modern ozone therapy. The device supports almost all currently known ozone applications.
Benefits of ozone
The main benefits of using ozone include its regenerative and anti-inflammatory effects. Ozone therapy relieves pain, improves tissue oxygenation, supports the immune system, and destroys bacteria, viruses, and fungi.
Application spectrum
-
Major autohemotherapy
-
Minor autohemotherapy
-
Rectal/Vaginal insufflation
-
Ozonation of liquids
-
Vacuum filling
Benefits of Ozone Therapy
Introduction to Major Autohemotherapy
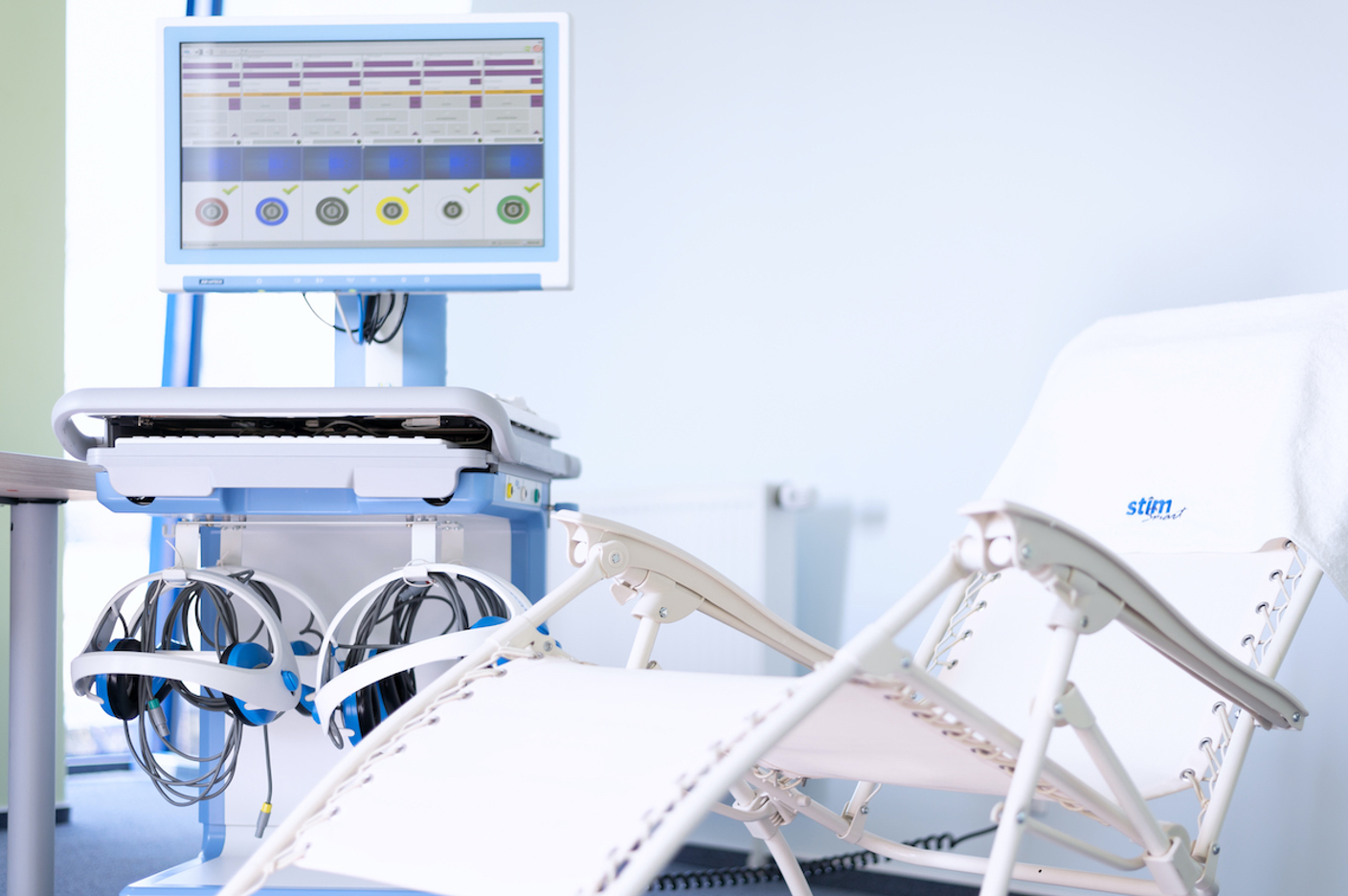
Stimsmart Therapy
Stimsmart therapy is one of the transcranial electrostimulation methods that adjusts and harmonizes disorders caused by both functional and organic diseases. During this non-invasive electrophysiological stimulation of the central nervous system, electrodes are placed on the client's head in a fronto-occipital position.
Clinical studies have shown that it is a completely safe method without side effects, which can be used alone or in combination with pharmacotherapy, and has long-term effects especially for patients suffering from diabetic polyneuropathy and microcirculation disorders.
Target Groups for Therapy
-
Neuropathic disorders
-
Chronic non-tumor pain
-
Fatigue syndrome, stress and body overload
-
Professional and recreational athletes
-
Plastic and cosmetic medicine
-
One-day orthopedics and surgery
-
Erectile dysfunction
-
Disorder of libido
-
Dermatology
E-Health and telemedicine
Telemedicine is a field encompassing healthcare activities, services, and systems delivered remotely through information and communication technologies, ranging from global health support and prevention to healthcare delivery and health system management.
Our company specializes in the development of telemedicine software.
Mobile applications for health insurance companies
Mobile applications of health insurance companies allow insured persons to access information about their health status as well as the health of their children. They also provide an overview of expenses for healthcare services received. The applications enable not only the display of an electronic insurance card, but also send reminders for vaccination dates, preventive check-ups, and similar appointments. In case of an emergency, the application makes it possible to call or send an SMS to a close contact.


Monitoring of medical devices
Software solution that does not replace qualified staff but helps make patient care more efficient.
Attending staff cannot be with all patients at the same time. We responded to this challenge by developing an e-platform that can monitor the current situation in individual rooms without compromising patient privacy.
Services for healthcare entities

Legislative support
Don’t know how to handle certification of your medical device?
We will provide you with regulatory support for its market introduction, including the preparation of complete technical documentation.
With our help, you will avoid non-compliances during audits.

Development
Would you like to develop a new functionality?
Based on your specific requirements, our experienced developers will create any software and hardware solution for your products.
Our experts are here for you.

Production
Lost a critical supplier?
We provide manufacturing of individual components with industrial-grade quality.
We operate production lines equipped with the latest technologies to maximize quality improvement and accelerate the entire manufacturing process.

Hotline
For any questions or reports of adverse effects, we are available at the following phone number:
+420 318 652 111
+420 377 438 111
To report medical device malfunctions, please contact us at:
service.medical@zat.cz.


Want to know more? Contact us!